Currently, the FDA allows biowaivers for Class I (high solubility and high permeability) and Class III (high solubility and low permeability) compounds of the Biopharmaceutics Classification System (BCS). Scientific evidence should be provided to support biowaivers for BCS Class I and Class III (high solubility and low permeability) compounds.
Abstract
Purpose
Methods
Data on the effects of excipients on drug permeability are needed to demonstrate that commonly used excipients do not affect the permeability of BCS Class III compounds, which would support the application of biowaivers to Class III compounds. This study was designed to generate such data by assessing the permeability of four BCS Class III compounds and one Class I compound in the presence and absence of five commonly used excipients.
Results
The permeability of each of the compounds was assessed, at three to five concentrations, with each excipient in two different models: Caco-2 cell monolayers, and in situ rat intestinal perfusion. No substantial increases in the permeability of any of the compounds were observed in the presence of any of the tested excipients in either of the models, with the exception of disruption of Caco-2 cell monolayer integrity by sodium lauryl sulfate at 0.1 mg/ml and higher.
Conclusion
The results suggest that the absorption of these four BCS Class III compounds would not be greatly affected by the tested excipients. This may have implications in supporting biowaivers for BCS Class III compounds in general.
Introduction
The Biopharmaceutics Classification System (BCS) is a framework for classifying drug substances based upon their aqueous solubility and permeability across biological membranes (). The FDA BCS guidance (2) provides recommendations for sponsors of investigational new drug applications (INDs), new drug applications (NDAs), abbreviated new drug applications (ANDAs), and supplements to these applications (SNDA) who wish to request a waiver of in vivo bioequivalence (BE) studies for immediate-release (IR) solid oral dosage forms. Drug substances are classified based on their intestinal permeability (or the fraction of oral dose absorbed) and aqueous solubility at multiple pH values covering the range found in the gastrointestinal (GI) tract. The solubility class boundary is based according to Papadopoulou et al. () the highest dose strength of the drug substance in a pharmaceutical product that is dissolved in 250 ml (8 oz.) of aqueous buffer.
Class I drugs have high permeability, and high solubility at all pH values between 1.0 and 7.5 (original FDA guidance from 2000), between 1.0 and 6.8 (new FDA draft guidance, May 2015) (2) or between 1.2 and 6.8 (European Medicines Agency (EMA) and World Health Organization (WHO) (4,5); such compounds are well absorbed but may have poor bioavailability (BA) due to extensive first-pass metabolism. Class II compounds have high permeability, but their solubility is below the class boundary at one or more pH values (e.g., at low pH for acids or at neutral pH for bases). The fraction absorbed may be limited by their solubility; therefore, it is not uncommon to see a wide range of extent of absorption for this class of compounds. Class III drugs have low permeability and high solubility, and their fraction absorbed is sometimes limited by their permeability. Class IV drugs have both low permeability and low solubility.
Class I drugs can be granted waivers from in vivo BE testing (“biowaiversâ€) due to their consistently high fraction absorbed, regardless of formulation (). Several authors have suggested that Class III compounds should also be eligible for biowaivers if a new formulation does not change the permeability or the GI transit time of the drug (–). WHO recommends, and the new FDA draft guidance (2) makes provision for, biowaivers for BCS Class III drug products, but only in cases where the generic and comparator drug products are very rapidly dissolving and the excipients meet certain criteria (e.g., qualitatively the same, quantitatively very similar, well-established for use in products containing that drug substance, do not affect GI motility or interactions with transport processes, and do not affect the pharmacokinetics (PK) of the drug substance) (2,5). This suggestion has also been proposed based upon a theoretical assessment of drug BA (,).
Certain excipients have been shown either to enhance the in vitro permeability of drugs (e.g., by changing membrane integrity or affecting transporters) or to modify GI transit time (,). Limited human data also suggest that some excipients may alter the BA of BCS Class III drugs (,). Several of these excipients have been demonstrated to alter membrane permeability directly, but others may work by inhibiting secretory transport mechanisms ().
In order to determine if a new formulation of a BCS III drug is suitable for a biowaiver, the potential effect(s) of the excipient(s) on permeability need to be evaluated. If no effect can be discerned in a well-validated in vitro model, it is reasonable to expect no change in permeability in vivo as well (–). First described for this purpose in 1989 (), the utility of the Caco-2 cell monolayer model for qualitative (e.g., rank-order or high vs. low) prediction of the oral absorption of drugs has been validated repeatedly since then (–).
This series of experiments had three objectives: 1) to determine whether a series of commonly used excipients would alter the permeability of model drug compounds in both a Caco-2 cell monolayer system (in vitro model) and an in situ rat intestinal perfusion model (surrogate for human in vivo data); 2) to determine whether there was a correlation between the Caco-2 and in situ rat intestinal perfusion models; and 3) to evaluate the relative suitability of the two models for testing the effects of excipients. The excipients were selected on the basis of their aqueous solubility and prevalence in IR formulations.
Five excipients were selected for testing: hydroxypropyl methylcellulose (HPMC), povidone, polyethylene glycol (PEG)-400, sodium lauryl sulfate (SLS), and lactose. All of these are commonly used in oral formulations, and some have been shown to act as penetration enhancers (). The model compounds evaluated for permeability were antipyrine (BCS Class I), acyclovir (Class III), atenolol (Class III), ganciclovir (Class III), and nadolol (Class III) (). The Class III compounds were selected because they are not substrates of P-gp and because they represent a range of the in vivo fraction absorbed, with atenolol > nadolol > acyclovir > ganciclovir (23). Antipyrine was included as a highly permeable control compound.
Materials and Methods
Materials
Ganciclovir was obtained from Toronto Research Chemicals (Toronto, Ontario, Canada). Acyclovir, antipyrine, atenolol, nadolol, digoxin, propranolol, d-glucose, d-lactose monohydrate, povidone K15, HPMC, PEG-400, SLS, 2-(N-morpholino)ethanesulfonic acid (MES), and Krebs Ringers buffer (KRB) were obtained from Sigma-Aldrich (St. Louis, MO, USA). Lucifer yellow (LY), Hanks’ balanced salt solution (HBSS), Dulbecco’s modified Eagle’s medium (DMEM), and 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) were obtained from Invitrogen (Carlsbad, CA, USA). Fetal bovine serum (FBS) was obtained from Omega (Tarzana, CA, USA). Penicillin, streptomycin, non-essential amino acids (NEAA), and trypsin-EDTA were obtained from CelGro (Herndon, VA, USA). Costar® tissue culture flasks and dual-chamber Transwell® plates were obtained from Corning (Corning, NY, USA).
The excipients used and their respective concentrations are given in Table ​TableI.I. The selection of the excipients and the concentrations used in the study were based on information obtained from the FDA’s Inactive Ingredient Database (24). The inclusion criteria for selection of the excipient and the concentrations included the frequency at which the excipient is used (they are all commonly used), the amount of excipient typically used, the solubility of the excipient in physiological fluids, and the concentration of the excipient in the commonly used dosing volume of 250 ml. This may be an important factor in determining the choice of excipients in new formulations.
Table I
Concentrations of Excipients Tested in the Two Permeability Models
| Excipient | Concentrations In Caco-2 (mg/ml) | Concentrations in rat intestinal perfusion (mg/ml) |
|---|---|---|
| Lactose | 0.024, 0.13, 0.24, 1.0, 2.0 | 0.024, 0.13, 0.24 |
| HPMC | 0.012, 0.036, 0.06, 1.0, 2.0 | 0.012, 0.036, 0.06 |
| PEG-400 | 0.015, 0.06, 0.18, 0.30 | 0.06, 0.18, 0.30 |
| Povidone | 0.024, 0.042, 0.06, 0.1, 0.2 | 0.024, 0.042, 0.06 |
| SLS | 0.01, 0.02, 0.04, 0.10, 0.17 | 0.01, 0.02, 0.04, 0.17 |
Bioanalysis
A liquid chromatography–tandem mass spectroscopy (LC-MS/MS) analytical method was developed and validated for the simultaneous determination of the five compounds (acyclovir, antipyrine, atenolol, ganciclovir, and nadolol) in the presence of five excipients (HPMC, povidone, PEG-400, SLS, and lactose). The analytical system was a PE SCIEX API 3000 LC-MS/MS with Flux Instruments Rheos 2000 pumps, a CTC Analytics autosampler, and an on-line degassing system. The LC column was a Thermo Aquasil C18 (3 μm, 50 × 2.1 mm) run at 300 μl/min with a 9-min gradient from 90% water/10% ammonium formate buffer (40 mM, pH 3.5) to 90% acetonitrile/10% ammonium formate buffer. Accuracy, based on analysis of replicate standards at 1 μM, was between 98.8 and 107% for all analytes, with coefficients of variation ranging from 2.6 to 5.9%. Similar accuracy and precision were found at lower concentrations.
Caco-2 Cells
Caco-2 cells (clone C2BBe1) were cultured in DMEM supplemented with 10% FBS, 1 mM sodium pyruvate, 100 μM NEAA, 2 mM L-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin. Stock cultures were grown in 175-cm2 flasks at 37°C in a humidified atmosphere containing 5% CO2. Cells were harvested with trypsin-EDTA, seeded at a density of 60,000 cells/cm2 on collagen-coated Transwell plates (0.4-μm pore size), and grown at 37°C in a humidified atmosphere containing 5% CO2. The culture medium was changed every other day for 10 days, and daily thereafter. Cell monolayers on Transwell plates were used for transport assays between 21 and 28 days post- seeding. Prior to use, each batch of monolayers was certified by measuring the transepithelial electrical resistance and the permeability of atenolol, propranolol, LY, and digoxin.
Permeability Across Caco-2 Cell Monolayers

Non-specific Binding and Recovery Assessment
Acyclovir (100 μM), antipyrine (10 μM), atenolol (100 μM), ganciclovir (100 μM), and nadolol (100 μM) were dosed together as a cassette in the apical chamber of a cell-free Transwell device (n = 3 replicates) to determine the non-specific binding (recovery) and cell-free apparent permeability across the membrane of each compound. Samples were collected from the donor and receiver chambers at multiple time points and analyzed by LC-MS/MS. “Cassette†refers to a small set of drugs (e.g., 3–5) or other compounds that are dosed together (co-dosed) for evaluation of their permeability (in vitro), absorption (in situ), or BA (in vivo). The assumption is that they do not interact with each other or interfere with each other’s absorption, in which case the co-dosed results should reflect the results that would be obtained if they were dosed individually.
Permeability Assay
The unidirectional (apical-to-basolateral (A→B)) and bidirectional (A→B and B→A) permeability of acyclovir, antipyrine, atenolol, ganciclovir, and nadolol, dosed as a cassette, was determined with Caco-2 cell monolayers. Bidirectional permeability was measured in the absence of excipients only (n = 3 replicates), whereas unidirectional permeability was measured both in the presence and absence of excipients (n = 4 replicates). The transport buffer was HBSS, supplemented with 10 mM MES or HEPES buffer and 10 mM D-glucose (15 mM D-glucose final concentration). The pH of the buffer in the apical (A→B donor, B→A receiver) chamber was 6.8, and the pH of the buffer in the basolateral (A→B receiver, B→A donor) chamber was 7.4. The apparent permeability (Papp) and recovery were calculated as follows:
dCr/dt is the rate of appearance of the compound in the receiver compartment (μM/sec); Vr and Vd are the volumes of the receiver and donor compartments, respectively (cm3); A is the surface area of the cell monolayer (1.13 cm2); C0 is the concentration (μM) of the dosing solution; and Cr final and Cd final are the concentrations (μM) in the receiver and donor compartments, respectively, at the end of the incubation period.
Permeability Across Rat Jejunum in Recirculating Intestinal Perfusion Model
Test System
Sprague–Dawley rats (300–350 g) were used to perform closed-loop, in situ intestinal perfusion. Rats were fasted for 12–18 h before the experiment, and water was supplied ad libitum.
Study Design
Four rats (n = 4) were used per treatment group, with a total of 17 groups: one control group (assay buffer, no excipients) and three or four treatment groups for each of the five excipients, with a different concentration for each group. The recirculating intestinal perfusion was performed with the five compounds dosed as a cassette (100 μM acyclovir, 10 μM antipyrine, 100 μM atenolol, 100 μM ganciclovir, and 100 μM nadolol) in KRB with 10 mM MES, pH 6.8.
Test Method
Rats were anesthetized with an intraperitoneal injection of ketamine/xylazine (1 ml/kg) and placed on a heating pad to maintain normal body temperature. After making a midline incision, the intestines were externalized. A 10-cm segment of the jejunum was tied off, cannulated at both ends to form a loop, and flushed with saline and air. The loop was equilibrated statically with dosing solution for 30 min, then flushed with air and perfused by recirculation (peristaltic pump) with 10 ml dosing solution at 0.2 ml/min. Samples of the perfusate were taken from the reservoir at 15, 30, 45, 60, 75, and 90 min for analysis by LC-MS/MS.
Sample and Data Analysis
The effective permeability coefficient, Peff (cm/s), was calculated as
where C is the concentration at a given time and C0 is the concentration in the dosing solution (μM); d(ln C/C0)/dt is the slope of a plot of the natural log of the concentration ratio in the perfusate vs. time (sec−1) at steady state, corrected for the apparent loss (due to dilution) of the impermeant marker FD-4 (fluorescein-conjugated dextran with a molecular weight of 4 kDa); V is the volume of the dosing solution (10 cm3); 2πrl is the luminal surface area (cm2), based on a radius (r) of 0.178 cm for the rat intestine () and the measured length of each intestinal segment, l (cm).
Results
LC-MS/MS
The LC-MS/MS method for acyclovir, antipyrine, atenolol, ganciclovir, and nadolol cassette analysis—in the presence and absence of the excipients HPMC, povidone, PEG-400, SLS, and lactose—had adequate sensitivity and selectivity and was valid for use in this study. The lower limit of quantification (LLOQ) was 0.005 μM, and the upper limit of quantification (ULOQ) was 1 μM for all analytes. The intra- and inter-assay accuracy and precision were within ±12% of nominal at all concentration levels in all three matrices (HBSSg, pH 7.4; HBSSg, pH 6.8; and KRB, pH 6.8). Benchtop, autosampler, and refrigerator stability were adequate, and no significant endogenous interference was found from HPMC, povidone, SLS, PEG-400, or lactose.
Bidirectional Permeability
As shown in Table ​TableII,II, all five compounds in the cassette had recoveries of at least 85% and high cell-free permeability under A→B test conditions. High recovery indicates little loss to either non-specific binding to the device or chemical instability during the assay, and high cell-free Pappindicates that the compounds were freely diffusible across the membrane (lack of aggregation, etc.).
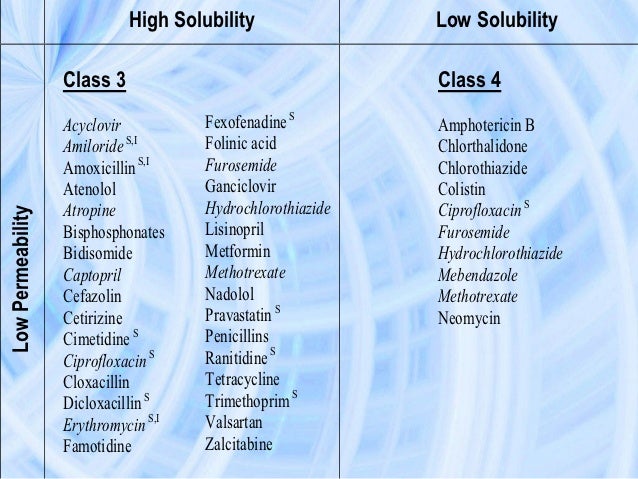
Table II
Non-specific Binding and Recovery of Antipyrine, Acyclovir, Atenolol, Ganciclovir and Nadolol from Cell-Free Permeability Test Devices
| Compound (μM) | Cell-free Papp (10−6 cm/s)a | Recovery (%) |
|---|---|---|
| Acyclovir (100) | 29.8 ± 1.8 | 86.1 ± 1.7 |
| Antipyrine (10) | 34.8 ± 0.8 | 85.8 ± 2.1 |
| Atenolol (100) | 33.8 ± 0.7 | 102 ± 1.1 |
| Ganciclovir (100) | 31.1 ± 1.4 | 92.3 ± 1.6 |
| Nadolol (100) | 33.0 ± 0.7 | 101 ± 1.5 |
Data presented as mean ± SD, n = 3
a For example, a value of “1.0†represents a Papp of 1.0 × 10−6 cm/s
The bidirectional permeability results presented in Table III demonstrate that the BCS Class I compound antipyrine had much higher permeability across Caco-2 cell monolayers than the other four model compounds in the cassette (all of which were BCS Class III compounds), with complete recovery and no significant efflux (efflux ratio was approximately equal to 1). The permeability of acyclovir, atenolol, ganciclovir, and nadolol were each quite low and similar in magnitude to each other; the efflux ratios ranged from 2.1 to 2.7.
Table III
Bidirectional Permeability and Recovery of Antipyrine, Acyclovir, Atenolol, Ganciclovir and Nadolol Across Caco-2 Cell Monolayers
| Compound (μM) | A→B | B→A | Efflux ratiob | ||
|---|---|---|---|---|---|
| Papp (10−6 cm/s)a | Recovery (%) | Papp (10−6 cm/s)a | Recovery (%) | ||
| Acyclovir (100) | 0.32 ± 0.14 | 82.2 ± 8.6 | 0.85 ± 0.11 | 94.9 ± 12.2 | 2.7 |
| Antipyrine (10) | 44.5 ± 0.70 | 104 ± 3.1 | 52.5 ± 3.80 | 98.8 ± 9.60 | 1.2 |
| Atenolol (100) | 0.29 ± 0.14 | 83.6 ± 9.2 | 0.62 ± 0.08 | 94.0 ± 11.0 | 2.2 |
| Ganciclovir (100) | 0.32 ± 0.16 | 82.9 ± 7.3 | 0.70 ± 0.09 | 92.7 ± 10.8 | 2.1 |
| Nadolol (100) | 0.29 ± 0.16 | 83.9 ± 9.1 | 0.71 ± 0.15 | 90.7 ± 12.2 | 2.5 |
Data presented as mean ± SD, n = 3
a For example, a value of “1.0†represents a Papp of 1.0 × 10−6 cm/s
b Efflux ratio is calculated from the mean Papp value in each direction
Unidirectional Permeability
The concentrations of the excipients tested are shown in Table ​TableI.I. The concentrations (in mg/ml) are shown with the two permeability models, Caco-2 cells and rat intestinal perfusion.
The results presented in Table ​TableIVIV demonstrate no consistent, concentration-dependent changes in the permeability of the model compounds across Caco-2 cell monolayers in the A→B (physiological absorptive) direction in the presence of up to 2.0 mg/ml lactose, 0.2 mg/ml povidone, 2.0 mg/ml HPMC, 0.04 mg/ml SLS, or 0.3 mg/ml PEG-400. The same was true for monolayer integrity (data not shown). On the other hand, SLS at concentrations of 0.1 and 0.17 mg/ml increased the post-experiment permeability of the monolayer integrity marker LY, indicative of damage to the cell monolayer (see footnote to Table ​TableIV).IV). The permeability of the model compounds in the presence of 0.1 mg/ml SLS was approximately 10-fold higher than control; permeability in the presence of 0.17 mg/ml SLS was not determined due to the evident cell monolayer damage.
Table IV
Effects of Excipients on In Vitro Caco-2 Cell Monolayer Permeability
| Excipient | Conc., mg/ml | Apparent permeability coefficient (Papp) (10−6 cm/s) | ||||
|---|---|---|---|---|---|---|
| Antipyrine | Acyclovir | Atenolol | Ganciclovir | Nadolol | ||
| Controla | 39.9 ± 2.14 | 0.28 ± 0.06 | 0.23 ± 0.05 | 0.25 ± 0.06 | 0.21 ± 0.06 | |
| Controlb | 43.6 ± 0.89 | 0.28 ± 0.06 | 0.25 ± 0.05 | 0.22 ± 0.05 | 0.21 ± 0.04 | |
| D-Lactose | 0.024a | 42.3 ± 5.53 | 0.18 ± 0.04 | 0.19 ± 0.05 | 0.14 ± 0.04 | 0.14 ± 0.04 |
| 0.13a | 41.6 ± 0.58 | 0.32 ± 0.11 | 0.57 ± 0.46 | 0.48 ± 0.42 | 0.44 ± 0.37 | |
| 0.24a | 39.8 ± 0.55 | 0.31 ± 0.20 | 0.27 ± 0.19 | 0.28 ± 0.19 | 0.27 ± 0.20 | |
| 1.0b | 33.3 ± 0.85 | 0.20 ± 0.05 | 0.16 ± 0.06 | 0.17 ± 0.05 | 0.14 ± 0.05 | |
| 2.0b | 33.1 ± 1.32 | 0.17 ± 0.14 | 0.14 ± 0.01 | 0.16 ± 0.01 | 0.13 ± 0.01 | |
| Povidone | 0.024a | 44.5 ± 0.91 | 0.17 ± 0.02 | 0.14 ± 0.01 | 0.15 ± 0.01 | 0.11 ± 0.01 |
| 0.042a | 40.4 ± 0.96 | 0.21 ± 0.03 | 0.15 ± 0.02 | 0.18 ± 0.03 | 0.13 ± 0.02 | |
| 0.06a | 41.9 ± 0.30 | 0.14 ± 0.004 | 0.11 ± 0.003 | 0.12 ± 0.002 | 0.09 ± 0.02 | |
| 0.1b | 38.5 ± 1.56 | 0.19 ± 0.05 | 0.13 ± 0.03 | 0.16 ± 0.04 | 0.12 ± 0.03 | |
| 0.2b | 36.3 ± 0.56 | 0.23 ± 0.06 | 0.16 ± 0.05 | 0.19 ± 0.05 | 0.15 ± 0.05 | |
| HPMC | 0.012a | 40.8 ± 0.99 | 0.15 ± 0.01 | 0.12 ± 0.01 | 0.14 ± 0.01 | 0.11 ± 0.01 |
| 0.036a | 37.3 ± 1.33 | 0.18 ± 0.04 | 0.14 ± 0.04 | 0.15 ± 0.04 | 0.14 ± 0.03 | |
| 0.06a | 39.2 ± 1.63 | 0.17 ± 0.05 | 0.14 ± 0.05 | 0.15 ± 0.05 | 0.14 ± 0.05 | |
| 1.0b | 33.2 ± 1.51 | 0.20 ± 0.06 | 0.15 ± 0.05 | 0.16 ± 0.05 | 0.13 ± 0.05 | |
| 2.0b | 33.5 ± 0.29 | 0.17 ± 0.03 | 0.13 ± 0.02 | 0.15 ± 0.03 | 0.11 ± 0.02 | |
| SLS | 0.01a | 46.9 ± 2.50 | 0.25 ± 0.07 | 0.24 ± 0.07 | 0.21 ± 0.07 | 0.23 ± 0.07 |
| 0.02a | 40.9 ± 1.11 | 0.34 ± 0.04 | 0.32 ± 0.05 | 0.30 ± 0.04 | 0.31 ± 0.05 | |
| 0.04a | 45.9 ± 0.56 | 0.35 ± 0.11 | 0.39 ± 0.12 | 0.30 ± 0.10 | 0.31 ± 0.10 | |
| 0.1a,c | 36.6 ± 1.23 | 2.48 ± 1.08 | 2.14 ± 0.96 | 2.23 ± 0.98 | 1.98 ± 0.87 | |
| 0.17 | Not determined due to disruption of monolayer integrity | |||||
| PEG 400 | 0.015b | 40.7 ± 0.96 | 0.34 ± 0.04 | 0.29 ± 0.04 | 0.30 ± 0.04 | 0.27 ± 0.04 |
| 0.06a | 38.9 ± 0.49 | 0.17 ± 0.01 | 0.17 ± 0.01 | 0.16 ± 0.01 | 0.13 ± 0.01 | |
| 0.18a | 37.2 ± 0.53 | 0.22 ± 0.06 | 0.18 ± 0.05 | 0.20 ± 0.05 | 0.14 ± 0.04 | |
| 0.30a | 37.2 ± 0.68 | 0.19 ± 0.06 | 0.16 ± 0.05 | 0.18 ± 0.05 | 0.12 ± 0.04 | |
Data presented as mean ± SEM (n = 4)
a,b Two separate sets of controls were run, each in parallel with a batch of test wells with excipients
c Integrity of Caco-2 cell monolayers was compromised (LY Papp 0.18 × 10−6 cm/s with buffer only; 0.30 × 10−6 cm/s with test compounds only (no excipients); 0.05 × 10−6 cm/s with 0.01 mg/ml SLS; 0.16 × 10−6 cm/s with 0.02 mg/ml SLS; 0.20 × 10−6 cm/s with 0.04 mg/ml SLS; 2.20 × 10−6 cm/s with 0.1 mg/ml SLS; 4.61 × 10−6 cm/s with 0.17 mg/ml SLS)
Permeability in the Recirculating In Situ Rat Intestinal Perfusion Model
As shown in Table ​TableVV and in Figs. Salman khan new hindi movie 2018. 1, ​,2,2, ​,3,3, ​,4,4, and ​and5,5, the permeability results in the recirculating in situ rat intestinal perfusion model also suggest that lactose (up to 0.24 mg/ml), povidone (up to 0.06 mg/ml), HPMC (up to 0.06 mg/ml), SLS (up to 0.17 mg/ml), and of PEG-400 (up to 0.3 mg/ml) do not have a substantial effect on the permeability of the model compounds across rat jejunum. The small sample size precludes definitive conclusions regarding statistical significance.
Table V
Effects of Excipients on In Situ Rat Jejunal Permeability
| Excipient | Conc., mg/ml | Effective permeability coefficient (Peff) (10−4 cm/s) | ||||
|---|---|---|---|---|---|---|
| Antipyrine | Acyclovir | Atenolol | Ganciclovir | Nadolol | ||
| Control | 1.43 ± 0.17 | −0.42 ± 0.24 | −0.33 ± 0.22 | −0.24 ± 0.17 | −0.41 ± 0.19 | |
| D-Lactose | 0.024 | 1.40 ± 0.20 | −0.51 ± 0.09 | −0.53 ± 0.09 | −0.44 ± 0.09 | −0.44 ± 0.08 |
| 0.13 | 1.28 ± 0.06 | −0.09 ± 0.36 | −0.17 ± 0.36 | −0.10 ± 0.19 | −0.21 ± 0.17 | |
| 0.24 | 2.03 ± 0.38 | −0.26 ± 0.22 | −0.16 ± 0.28 | −0.11 ± 0.17 | −0.17 ± 0.24 | |
| Povidone | 0.024 | 1.63 ± 0.34 | 0.16 ± 0.19 | 0.21 ± 0.17 | 0.05 ± 0.15 | 0.13 ± 0.015 |
| 0.042 | 1.15 ± 0.07 | −0.22 ± 0.24 | −0.20 ± 0.21 | −0.10 ± 0.18 | −0.11 ± 0.16 | |
| 0.06 | 1.33 ± 0.42 | 0.03 ± 0.14 | 0.12 ± 0.15 | −0.25 ± 0.09 | −0.23 ± 0.08 | |
| HPMC | 0.012 | 1.43 ± 0.21 | 0.35 ± 0.18 | 0.36 ± 0.19 | 0.10 ± 0.17 | 0.07 ± 0.17 |
| 0.036 | 0.91 ± 0.19 | 0.05 ± 0.06 | 0.00 ± 0.07 | −0.04 ± 0.03 | −0.12 ± 0.03 | |
| 0.06 | 1.68 ± 0.35 | 0.21 ± 0.14 | 0.06 ± 0.03 | 0.27 ± 0.07 | 0.40 ± 0.07 | |
| SLS | 0.01 | 1.75 ± 0.23 | 0.19 ± 0.012 | 0.46 ± 0.13 | 0.23 ± 0.15 | 0.32 ± 0.15 |
| 0.02 | 0.93 ± 0.26 | 0.41 ± 0.07 | 0.46 ± 0.09 | 0.36 ± 0.07 | 0.37 ± 0.10 | |
| 0.04 | 1.08 ± 0.18 | −0.05 ± 0.07 | 0.02 ± 0.05 | 0.00 ± 0.01 | 0.01 ± 0.02 | |
| 0.17 | 1.57 ± 0.46 | −0.38 ± 0.14 | −0.30 ± 0.17 | −0.28 ± 0.14 | −0.36 ± 0.10 | |
| PEG 400 | 0.06 | 1.17 ± 0.22 | 0.17 ± 0.07 | 0.29 ± 0.07 | 0.26 ± 0.07 | 0.24 ± 0.08 |
| 0.18 | 0.90 ± 0.26 | 0.27 ± 0.14 | 0.27 ± 0.13 | 0.30 ± 0.09 | 0.25 ± 0.08 | |
| 0.30 | 0.82 ± 0.15 | 0.07 ± 0.02 | 0.09 ± 0.03 | 0.02 ± 0.05 | 0.01 ± 0.01 | |
| 0.30a | 1.53 ± 0.18 | −0.21 ± 0.19 | −0.13 ± 0.20 | −0.24 ± 0.13 | −0.23 ± 0.13 | |
Data presented as mean ± SEM (n = 4)
aSecond run
Pappvs.d-Lactose concentration.
Pappvs. Povidone concentration.
Pappvs. HPMC concentration.
Pappvs. SLS concentration.
Pappvs. PEG-400 concentration.
The permeability data were noticeably less variable in the Caco-2 model than in the in situ rat perfusion model: median relative standard deviation for antipyrine of 2.64% in the Caco-2 model vs. 18.5% in the rat perfusion model; a range of 17.0 to 19.4% for the Class III compounds in the Caco-2 model vs. 46.4 to 65.2% in the rat perfusion model.
Discussion
An LC-MS/MS method was validated for acyclovir, antipyrine, atenolol, ganciclovir, and nadolol determinations as a cassette in the absence and presence of excipients. This approach allowed for a relatively rapid assessment of the results of both arms of the study. This approach may be used to test the effects of other excipients on the same model compounds.
The permeability of all five model compounds across intestinal epithelia was measured both in vitro (Caco-2 cell monolayers) and in situ (rat intestinal perfusion). One problem with the intestinal perfusion model is that negative Peff values are often observed for low-permeability compounds. The most likely reason for a negative Peff value is that net water flux cannot be assessed accurately. When a small amount of a supposedly “impermeable†marker (FD-4 in this case, used to correct for water secretion) is absorbed, mathematically it results in overcorrection for water secretion, resulting in negative Peff values for low-permeability compounds. Currently, there are no perfect markers for the rat intestinal perfusion model. FD-4 was used as the impermeable marker in this study, since this marker is widely accepted in the literature. Although negative Peff values for low-permeability compounds suggest that the rat perfusion model may be variable and potentially insensitive to small changes in drug absorption, this system is widely accepted for the evaluation of permeability.
The permeability of the BCS Class I compound antipyrine across Caco-2 cell monolayers was high in both directions, with no asymmetry (ratio of B→A Papp to A→B Papp near unity). The Caco-2 A→B (absorptive direction) permeability of the Class III model compounds acyclovir, atenolol, ganciclovir, and nadolol were all very low and nearly identical in magnitude (between 0.29 × 10−6 and 0.32 × 10−6 cm/s). The efflux ratios of the Class III compounds ranged from 2.1 to 2.7, a moderate degree of apparent efflux that is due in part to the fact that, in each case, the recovery was somewhat lower in the A→B direction than in the B→A direction, which would tend to underestimate the Papp in the A→B direction (the denominator in the efflux ratio), leading to an overestimate of efflux. Clinically, linear peak plasma concentration as a function of oral dose (PK) has been reported for atenolol () and nadolol (), suggesting no significant interaction with efflux transporters. For acyclovir () and ganciclovir (), decreasing exposure as a function of dose has been reported after oral administration; this is evidence for possible involvement of active intestinal uptake and is not consistent with an interaction with efflux transporters.
With the exception of an effect of SLS on Caco-2 cell monolayers at concentrations of 0.1 and 0.17 mg/ml, where cell monolayer integrity was clearly compromised, none of the tested excipients caused a substantial increase in the permeability of any of the model compounds in either model. The effect of SLS on Caco-2 cell monolayer integrity at higher test concentrations was clearly a model-specific toxic effect, and therefore may not be pharmaceutically relevant. Other studies have reported positive effects of excipients on the permeability of different drugs, but the effects appear to be dependent on the excipient and its concentration (,–). For example, sorbitol and SLS reduce the bioavailability of the Class I drug risperidone, the former due to increased GI motility and the latter by an unknown mechanism (). With the exception of SLS at the two highest tested concentrations, the excipients used in this study do not appear to disrupt cell monolayer integrity at the concentrations used, whereas some of the excipients used in other studies (e.g., sodium caprate ()) may be more likely to do so.
This study focused on the effects of individual excipients on permeability and did not evaluate the effects of combining multiple excipients (which would be commonly done in an IR formulation) on permeability. Because of the vast number of excipient combinations and concentration combinations, testing for the synergistic effects of combinations was not feasible.
While the limitations of the Caco-2 cell monolayer model (like any other preclinical model) are appreciated by investigators in the field, the FDA does accept Caco-2 data for BCS classification. In spite of its limitations, based on the fact that Caco-2 permeability data have lower variability between replicates than data obtained from in situ recirculating rat intestinal perfusion, the Caco-2 cell monolayer model may be more sensitive in terms of detecting effects of an excipient on drug permeability. On the other hand, Caco-2 cell monolayers may be overly sensitive to some excipients, as with SLS at or above 0.1 mg/ml. Therefore, a positive result may need to be followed up in vivo to rule out a false positive.
Except for SLS at concentrations at or above 0.1 mg/ml (at which concentrations the effects are on monolayer integrity rather than due to permeability enhancement), the results of this study with a set of four BCS Class III model compounds support the more general hypothesis that these five commonly used excipients do not affect the permeability of BCS Class III compounds. Biowaivers for Class III compounds should be considered when excipients used in a formulation have been shown not to affect permeability. There are many recent examples of biowaivers granted by the WHO for Class III drugs (–), as well as the assessment that BE of many of the IR drug products available today can be assured with an in vitro dissolution test (). The results of this study support biowaivers for BCS Class III compounds. This would reduce the amount of time needed to get new formulations to patients who need them, reduce the cost of developing new formulations, and reduce the cost of modifying or improving the manufacturing process for products containing Class III drugs. From an ethical point of view, it also reduces the needless exposure of healthy volunteers to drugs without therapeutic benefit.
Conclusion
The influence of five excipients (lactose, povidone, HPMC, SLS, and PEG400), commonly used in IR formulations, on the permeability of five model drugs (acyclovir, antipyrine, atenolol, ganciclovir, and nadolol) was investigated. Two test systems were used: Caco-2 cell monolayers and in situ recirculating rat intestinal perfusion; both have been used for absorption enhancement studies. Within the tested concentration range for all drug–excipient combinations, the excipients caused no substantial increases in drug permeability, except in the case of SLS at concentrations (0.1 mg/ml and higher) where cell monolayer integrity was compromised. These results should support biowaivers for BCS Class III (high solubility–low permeability) compounds, but only for compounds similar in permeability to those tested and which have been formulated with the excipients used in this study.
ACKNOWLEDGMENTS AND DISCLOSURES
The authors extend special thanks to the Product Quality Research Institute (PQRI) and Absorption Systems for their financial support of this work and declare no potential conflicts of interest.
Disclaimer
The views expressed in this paper are those of the authors and should not be interpreted as those of the FDA.
ABBREVIATIONS
| BA | Bioavailability |
| BCS | Biopharmaceutics classification system |
| BE | Bioequivalence |
| IR | Immediate-release |
| KRB | Krebs Ringers buffer |
| LC-MS/MS | Liquid chromatography-tandem mass spectroscopy |
| LY | Lucifer yellow |
| MES | 2-(N-morpholino)ethanesulfonic acid |
| NEAA | Non-essential amino acids |
| Papp | Apparent permeability coefficient |
| PEG | Polyethylene glycol |
| PK | Pharmacokinetics |
| SD | Standard deviation |
| SEM | Standard error of the mean |
References
Abstract
BCS and BDDCS are complimentary, not competing, classification systems that aim to improve, simplify, and speed drug development. Although both systems are based on classifying drugs and NMEs into four categories using the same solubility criteria, they differ in the criterion for permeability and have different purposes. Here the details and applications of both systems are reviewed with particular emphasis of their role in drug development.
INTRODUCTION
The FDA’s Biopharmaceutics Classification System(BCS)1 is based on the work of Amidon and coworkers with the core idea being that in vitro methodology, centrally embracing permeability and solubility, with qualifications related to pH and dissolution, may qualify drug products for a waiver of in vivo bioequivalence studies. The objective of the BCS is to predict in vivo performance of drug products from in vitro measurements of permeability and solubility.
In 2005, Wu and Benet recognized that for drugs exhibiting high intestinal permeability rates the major route of elimination in humans was via metabolism, while drugs exhibiting poor intestinal permeability rates were primarily eliminated in humans as unchanged drug in the urine and bile. They proposed that a biopharmaceutics drug disposition classification system (BDDCS) could serve as a basis for predicting the importance of transporters in determining drug disposition, as well as in predicting drug-drug interactions.
The major differences between BCS and BDDCS relate to their purpose and the measurement for classification as depicted in Table 1. The purpose of BCS is to characterize drugs for which products of those drugs may be eligible for a biowaiver of in vivo bioequivalence studies. The purpose of BDDCS is to predict drug disposition and potential drug-drug interactions in the intestine and the liver, and potentially the kidney and brain. Both BCS and BDDCS use solubility as one of the two classification criteria. The solubility parameter utilized may be called the FDA solubility, that is, an estimate of the ability of the drug at its highest dose strength to completely dissolve in 250 ml of water over a pH range between 1 and 7.5 at 37°C. For a drug to be considered highly soluble in the two classification systems, the drug from its highest strength regulatory approved dosage form must go completely into solution at its lowest solubility over this pH range in 250 ml of water. As we have recently noted, FDA solubility is a property of the drug in a formulation and is not an intrinsic property of the active pharmaceutical ingredient itself. The second classification parameter, and where the two systems differ, is related to intestinal permeability. In BDDCS, predictions are based on intestinal permeability rate, which was found to be related to extent of drug metabolism. In BCS, biowaivers are based on the extent of intestinal absorption, which in a number of cases does not correlate with intestinal permeability rate.
Table 1
| BDDCS | BCS |
|---|---|
| Purpose | |
| Predicting drug disposition and drug-drug interactions in the intestine and liver | Facilitate biowaivers of in vivo bioequivalence studies |
| Criterion | |
| Predictions are based on intestinal permeability rate | Biowaivers are based on extent of intestinal absorption (permeability), which in a number of cases does not correlate with rate of jejunal permeability. |
THE BCS AND ITS USE IN DRUG DEVELOPMENT
The BCS characterizes drugs into four classes according to their FDA solubility and permeability as depicted in Fig. 1. In 2000, the FDA promulgated the BCS system as a science-based approach to allow waiver of in vivo bioavailability and bioequivalence testing of immediate-release solid oral dosage forms for Class 1 high solubility, high permeability drugs when such drug products also exhibited rapid dissolution1. This waiver is based on a triple-tier rationale where: a) high solubility insures that drug solubility will not limit dissolution, and thus absorption, b) high permeability insures that drug is completely absorbed during the limited transit time through the small intestine, and c) rapid dissolution insures that the gastric emptying process is the rate-limiting step for absorption of highly soluble and highly permeable drugs. Drug sponsors are allowed to use mass balance, absolute bioavailability, or human intestinal perfusion studies to demonstrate high permeability1. The FDA Guidance, however, also recommends possible methods not involving human subjects including in vivo or in situ intestinal perfusion in a suitable animal model, and/or in vitro permeability methods using excised intestinal tissues or monolayers of suitable epithelial cells1,, usually the Caco-2 cell system. However, some studies have shown that in vitro cellular permeability criteria recognized in the FDA’s BCS guidance may not always correctly predict the extent of drug absorption in humans–.
The Biopharmaceutics Classification System (BCS) as defined by the FDA1 after Amidon et al.
In 2010, the European Medicine’s Agency (EMA) revised its bioequivalence guideline stating that demonstration of complete absorption in humans is preferred for biowaiver of BCS Class 1 drug applications rather than measures of high permeability9. The criteria for complete absorption in the EMA Guidline is ≥85% measured extent of absorption in humans based either upon absolute bioavailability or mass balance studies9.
The correlation between intestinal permeability rate and the extent of absorption in humans came from the results of in vivo studies with 34 drugs and endogenous substances, where a good correlation was observed between jejunal permeability from human intestinal perfusion studies and the fraction of the oral dose absorbed in humans. However, in these early human intestinal perfusion studies, no drugs were investigated that subsequently showed a discordance between cellular system permeability rates and the extent of absorption in humans. It is now generally recognized by the FDA, the EMA and the research scientists in the field that high cellular permeability rates do correctly predict a high extent of absorption. Discordance is essentially only found for some non-metabolized drugs exhibiting low cellular permeability rates but complete absorption.
The role of BCS in drug development is facilitating the possibility of obtaining a waiver of in vivo bioequivalence studies for drug products, where the regulatory agencies recognize the drug as BCS Class 1 and where the dissolution rate of the new drug product meets the rapid dissolution criteria of the regulatory agencies. This is definitely a simplifying and cost saving procedure in drug development. However, there is no predictive benefit to BCS. Studies in humans must be carried out to show that the drug achieves complete absorption (≥90% for FDA and ≥85% for EMA, although the FDA has indicated informally that ≥85% may be appropriate for a biowaiver). Yet, as stated above, cellular studies exhibiting high permeability rates can give sponsors confidence that a high solubility compound will meet the extent of absorption criteria of the regulatory agencies prior to obtaining actual extent of absorption measures in humans. Since the promulgation of the FDA BCS Guidance in 2000, a number of new possible class boundaries have been proposed for additional biowaivers. For example, the EMA will grant biowaivers for BCS Class 3 drugs with high solubility but limited absorption9. In addition, a World Health Organization (WHO) Technical Report11 suggests that biowaivers may be appropriate for so-called BCS Class 2a drugs, weak acids that exhibit low solubility only at low pH. The WHO recommended criteria for such drug products would be rapid dissolution at pH 6.8 and a similar dissolution profile to the innovator product at pH 1.2, 4.5 and 6.8. Like the EMA, the WHO Technical Report also recommends biowaiver eligibility for Class 3 very rapidly dissolving drug products that contain no inactive ingredients that are known to alter GI motility and/or absorption11. However, at this time only BCS Class 1 drugs are eligible for a biowaiver of in vivo bioequivalence from the FDA1 and BCS Class 1 as well as some Class 3 drugs by the EMA9.
BDDCS
As described above the purpose of BDDCS is to predict drug disposition and potential drug-drug interactions in the intestine and the liver with an emphasis on defining which drugs would be amenable to enzymatic-only and transporter-only disposition and drug-drug interactions, as well as where transporter-enzyme interplay may be important. Recent reviews from the Benet Lab– have defined these enzymatic, transporter and transporter-interplay characteristics with potential transporter effects following oral dosing as depicted in Figure 2. The recognition of the correlation between BCS intestinal permeability and BDDCS extent of metabolism by Wu and Benet preceded an explanation for these findings. We hypothesize now that high permeability rate compounds are readily reabsorbed from the kidney lumen and from the bile, facilitating multiple access to the metabolic enzymes. For example, consider the BCS/BDDCS Class 1 drug letrozole. This completely oral available drug is primarily eliminated by metabolism via CYP3A4 and CYP2A6 enzymatic processes with less than 4% of the dose excreted unchanged in the urine. However, letrozole is only 60% bound to plasma proteins and thus it might be expected, based on glomerular filtration rate and fraction unbound, that renal clearance could approach 48ml/ml. Yet the total clearance for letrozole is only 40.5 ml/min with less than 4% excreted unchanged. Thus, this high permeability compound is reabsorbed in the kidney tubules (and possibly from the bile) with the major route of elimination being metabolic processes. The rationale for the correlation between intestinal permeability rate and the extent of metabolism appears to be based on the fact that high permeability rate compounds are reabsorbed from potential unchanged drug excretion routes in the body and thus can only be eliminated through metabolism. This hypothesis, then led us to conclude that the measure of high permeability rate in making the BDDCS assignment need not necessarily be a human biological membrane or membrane surrogate, but that passive permeability in any appropriate membrane model may provide the correct assignment. This topic will be discussed further below.
Transporter effects predicted by BDDCS following oral dosing.
THE USE OF BDDCS FOR DRUGS ALREADY ON THE MARKET
Table 2 lists six potential uses of BDDCS in characterizing drugs that have already reached the market. The text below provides greater detail for each of these six potential uses.
Table 2
|
For drugs already on the market, BDDCS provides potential predictability of drug-drug interactions that had not been anticipated or tested in the drug approval process. For example, our laboratory recognized that atorvastatin was a BDDCS Class 2 drug exhibiting extensive metabolism and poor solubility. Thus, as shown in Fig. 2, we recognized that atorvastatin may potentially exhibit a drug-drug interaction with inhibitors of hepatic uptake transporters. We first demonstrated in cellular and isolated perfused rat liver studies that atorvastatin, as well as its two active hydroxylated metabolites, were substrates for human and rat OATPs and that inhibition of OATP uptake would decrease atorvastatin metabolism. We then carried out whole animal studies in rats to confirm this finding in intact animals. We then demonstrated in healthy volunteers that a single intravenous dose of rifampin, a potent OATP inhibitor would significantly increase the total AUC of atorvastatin acid by 6.8±2.4-fold and that of 2-hydroxy-atorvastatin acid and 4-hydroxy-atorvastatin acid by 6.8±2.5-fold and 3.9±2.4-fold, respectively. Of course, once recognizing that atorvastatin and its active metabolites are substrates for OATP1B1, then it would logically follow that genetic variants in this transporter would affect atorvastatin pharmacokinetics, as has been demonstrated. Thus, BDDCS is also useful in predicting where pharmacogenetic variants can yield meaningful drug disposition changes.
In a further study we demonstrated that inhibiting the hepatic uptake transporter for glyburide would also significantly increase its area under the curve and that blood glucose levels were lower than those observed after dosing with glyburide alone. Glyburide is primarily a substrate of OATP1B3, which does not exhibit significant changes in activity with genetic variants, and therefore one would suspect that a pharmacogenetic study of the transporter would not yield significant changes. However glyburide is a substrate for CYP2C9, with known disposition changes for the genetic variants of this enzyme.
It is important to recognize that the BDDCS characterization of transporter effects, and transporter enzyme interplay as depicted in Fig. 2 does not predict that every drug in each class will display the effects listed. Rather, BDDCS predicts what transporter effects may occur, and which may not, and what should be tested. As an example, the Class 2 drug felodipine is a CYP3A substrate, but not a substrate for P-glycoprotein, and we used it as our control in examining Class 2 drug efflux transporter-enzyme interplay. Furthermore, one cannot be sure that a cellular transporter-enzyme interaction will translate into an in vivo clinically relevant interaction, even when the in vitro Ki values suggest that the interaction needs to be tested. As an example, numerous publications concerning the pharmacogenomics of warfarin have shown that accounting for the genetic variants of CYP2C9 and VKORC1 plus other patient parameters can only explain about 55% of the variability observed for this drug in patient populations. Since warfarin is a Class 2 drug, we asked could it be a substrate for an uptake transporter, and if so might knowledge of this transporter genotype increase the predictability? Rat and human hepatocyte studies showed that warfarin appeared to be a substrate for OATP uptake that could be inhibited by rifampin, which might account for an ~30% change in AUC. The in vitro interaction was of the same magnitude as what we had observed in vitro for glyburide. We then carried out a human study that showed that there was no significant increase in warfarin blood concentrations in the presence of the OATP inhibitor rifampin.
Recently the FDA has recommended that studies in renal failure patients be carried out even for drugs where renal elimination of unchanged drug is minimal23. This recommendation comes about in part based on a finding that was related to the development and characterization of BDDCS. Previous studies of changes in drug metabolism in renal failure patients for drugs primarily eliminated by hepatic metabolism were thought to be related to the effects of uremic toxins as either potential inhibitors or down regulators of metabolic enzymes. However, this could be tested in vitro and was shown not to occur in many cases. We began to recognize that previously unexplained effects of renal disease on hepatic metabolism can result from accumulation of substances (toxins) in renal failure that modify hepatic uptake and efflux transporters–, and that this mechanism could explain why BDDCS Class 2 drugs could demonstrate changes in metabolism in renal failure, while this would not be observed for BDDCS Class 1 drugs when in vitro uremic toxins did not alter microsomal metabolism.
To demonstrate the relevance of the potential effect of uremic toxins in patients we compared the pharmacokinetics of oral and IV erythromycin in patients with end stage renal disease versus healthy volunteers. Erythromycin is a BDDCS Class 3 drug that is primarily eliminated unchanged in the bile. It is a substrate for hepatic uptake transporters that we had previously shown can be inhibited by uremic toxins. We demonstrated that the hepatic clearance of erythromycin in end stage renal disease patients was decreased by 31% (p=0.01) and that bioavailability was increased 36%. Since we had given the drug both intravenously and orally, we calculated that there was no change in the fraction of the oral dose absorbed multiplied by the potential gut availability (Fabs·Fg). This would be expected even though erythromycin, a Class 3 BDDCS drug is a substrate for an intestinal uptake transporter, since it is not possible for the uremic toxins to be present in the intestine. Thus the BDDCS allows investigators to predict the potential effect of uremia on hepatic metabolism and biliary excretion.
To facilitate use of the BDDCS system for making predictions for drugs on the market, we recently compiled the BDDCS classification for 927 drugs, which include 30 active metabolites. Of the 897 parent drugs, 78.8% (707) are administered orally. Where the lowest measured solubility was found in the literature this value was reported for 72.7% (513) of these orally administered drugs. Measured values are reported for the percentage excreted unchanged in the urine, Log P and Log D 7.4, when available. For all 927 compounds the in silico parameters for predicted Log solubility in water, calculated Log P, Polar Surface Area and the number of hydrogen bond acceptors and hydrogen bond donors for the active moiety are also provided, thereby allowing comparison analyses for both in silico and experimentally measured values. We showed that when comparing the in silico parameters across the four classes, there is a distinct difference between Class 2 and Class 3 compounds. However, surprisingly the Log P and solubility in silico parameters for the Class 1 drugs appear to be intermediate between those for Class 2 and Class 3 and not very different than the parameters for the Class 4 drugs. We note this failure of in silico parameters to efficiently predict whether a drug will be Class 1, believed by many to be the most desirable due to high solubility and high permeability, versus Class 4 drugs that are low solubility and low permeability.
Most recently we have shown that the prediction of brain disposition of orally administered drugs may be improved using BDDCS. It is generally believed that high Log P, high permeability and lack of P-gp efflux are desirable characteristics for CNS drug candidates to become marketed CNS drugs–. From the literature we were able to identify 153 marketed drugs that met three criteria: a) central or lack of central pharmacodynamics effects were known; b) the BDDCS class was identified; and c) information was available as to whether the drug was or was not a substrate for P-glycoprotein. About 98% of BDDCS Class 1 drugs were found to be markedly distributed throughout the brain; this includes 17 BDDCS Class 1 drugs known to be P-gp substrates. Thus, we expand upon the transporter effects listed in Fig. 2 for Class 1 drugs. We now believe that transporter effects are minimal, and clinically insignificant, for Class 1 drugs in the gut, liver and brain, and suspect that this is also true for the kidney. Recognition that BDDCS Class 1 drugs that are P-gp substrates will still yield central effects allowed us to decrease the number of compounds incorrectly predicted from in silico parameters in terms of disposition from the 19–23% employing previous methods,, to less than 10%. This finding of a lack of a clinically significant effect of P-gp on brain disposition of BDDCS Class 1 drugs has marked implications for predicting drug disposition and effects of NMEs, as will be described in the last section of this report.
Finally, there is also an application to BCS biowaivers inherent in the BDDCS Classification System. Benet and coworkers recognized that for 29 drugs where measured human intestinal permeabilities were available, the extent of metabolism correctly predicted high versus low permeability for 27 of 29 (or 93%) of the measured human intestinal permeabilities in terms of BCS. Thus, Benet and colleagues recommended that regulatory agencies add the extent of drug metabolism (i.e., ≥90% metabolized for FDA and ≥85% metabolized for EMA) as an alternate method for determining the extent of drug absorption in defining Class 1 drugs suitable for a waiver of in vivo studies of bioequivalence. The authors propose that the following criteria be used to define the ≥90% metabolized for FDA marketed drugs: “Following a single oral dose to humans, administered at the highest dose strength, mass balance of Phase I oxidative and Phase II conjugative drug metabolites in the urine and feces measured as either unlabeled, radioactive labeled or non-radioactive labeled substances, account for ≥90% of the drug dose. This is the strictest definition for a waiver based on metabolism. For an orally administered drug to be ≥90% metabolized by Phase I oxidative and Phase II conjugative process it is obvious that the drug must be absorbed.†In their 2010 revised bioequivalence guidance9, EMA incorporated this recommendation. FDA scientists have also supported this recommendation, although no formal written guidance change has been issued.
THE ROLE OF BDDCS IN THE DRUG DEVELOPMENT OF NEW MOLECULAR ENTITIES (NMEs)
Although BDDCS can be used for characterizing disposition of drugs already on the market, as detailed in Table 2 and in the text above, the goal of BDDCS was to predict and characterize drug disposition for new molecular entities. Table 3 lists those uses and the text below is a prescription for utilizing BDDCS with NMEs.
Table 3
Additional Uses of BDDCS for New Molecular Entities and its Role in Drug Development
|
For an NME it would be most useful to predict its BDDCS class prior to any studies in humans, animals or even cellular systems. The recognition of the correlation between intestinal permeability rate and extent of metabolism allows prediction of BDDCS class for an NME to be based on passive membrane permeability35. Initially we proposed to follow the BCS permeability rate measure in the Caco-2 cellular system using metoprolol, but now more preferably labetalol, as the cutoff between high and low permeability compounds. However, since we now believe that it is the passive permeability that is the predictive parameter, we suggest that even studies with an artificial membrane such as PAMPA will provide a reasonable prediction of BDDCS Class 1 and 2 versus BDDCS Class 3 and 4 using labetalol as the cut off marker. We evaluated35 the permeability results for 21 drugs studied by three different PAMPA models (a lipid/oil/lipid tri-layer, a bio-mimetic, and a hydrophilic filter membrane PAMPA assay). For these 21 drugs the human extent of absorption and metabolism was known. In this evaluation the extent of absorption or metabolism was defined as being low or high if it was less than 30% or ≥90%, respectively. Permeability was defined as being low or high if it was less than 2.0 or ≥3.5x10−6cm/s, respectively based on a marked differentiation using these cut-offs for 19 of the 21 drugs. The high PAMPA permeability for all three models accurately predicted BCS ≥90% absorption very well and only slightly less accurately BDDCS metabolism. However, for low PAMPA permeability while the system very accurately predicted poor BDDCS metabolism the systems only correctly predicted absorption 25% of the time. Based on these data we suggest that passive transcellular drug permeability in an artificial membrane may reasonably predict extensive versus poor human metabolism35. Note that we are not suggesting that the permeability rate cut offs listed above are numbers that may be translated to other laboratories. They are just the values from our in vitro permeability rate analyses used to attempt to differentiate the 21 drugs for which human in vivo permeability rate measures and extent of metabolism have been reported.
Although we believe it is quite easy, and using high throughput methods, to predict with good accuracy Class 1 and Class 2 versus Class 3 and Class 4 assignment of an NME, it is not easy to differentiate high solubility from low solubility. The major impediment is that the BDDCS (and BCS) solubility criterion is based on the highest marketed dose strength. Of course, for an NME such knowledge may be years away from the compounds initial evaluation. Therefore we propose following the recommendation of Pfizer scientists to use a solublity cutoff of 200μg/ml (i.e., 50 mg highest dose strength) in the initial evaluation. Thus, compounds with a lowest solubility over the pH range 1–7.5 being greater than 200μg/ml would be assigned Class 1 or Class 3 and those with a lowest solubility less than 200μg/ml would be assigned Classes 2 and 4. As we have noted previously, in silico predictions of solubility are not very reliable and thus we also recommend following the Pfizer protocol of utilizing a high throughput equilibrium solubility assay for NMEs in simulated gastric fluid at pH 1.2 and in 50 mM phosphate buffer pH 6.5, for the initial assignment. Thus, very early in the drug development of an NME, using only in vitro methods to assign BDDCS class, sponsors will be able to predict the major route of elimination of an NME in humans (metabolism versus excretion of unchanged drug in the urine and bile) and predict the relevance of transporters and transporter-enzyme interplay in drug disposition. If an NME is BDDCS Classes 1 or 2, the drug should exhibit good absorption, but not necessarily good bioavailability. In contrast, if the NME is BDDCS Classes 3 or 4, then good absorption will only be achieved if the NME is a substrate for an intestinal uptake transporter or possibly small enough to pass through intestinal pores.
There is a marked difference in the BDDCS class distribution of drugs on the market as opposed to NMEs. We previously estimated the distribution for NMEs based on all recently synthesized medicinal compounds. In Fig. 3 we depict the distribution of oral immediate release drugs on the market versus small molecule NMEs, the latter percentages determined from a data set of Professor Oprea37 encompassing 28,912 medicinal chemistry compounds tested for at least one target and having affinities of μM or less concentrations. While 40% of oral immediate release marketed drugs are Class 1, only 18% of NMEs fall in this category. This difference is primarily related to Class 2 drugs, where 33% of marketed oral immediate release products are found versus 54% of NMEs. As can be seen in Fig. 3 quite similar numbers between marketed drugs and NMEs are found for Classes 3 and 4. That is, in essence, NMEs are becoming larger, more lipophilic and less soluble, with time in the drug discovery paradigm.
Distribution of 698 oral immediate release drugs on the market and NME percentages from a data set of 28,912 medicinal chemistry compounds tested for at least one target and having affinities at μM or less concentrations37 using BDDCS criteria.
Early characterization of the BDDCS class of an NME offers information related to of all those characteristics listed in Table 2 for drugs on the market. Of particular relevance, early in drug development, may be the information related to potential brain distribution as described in our recent publication. That is, if central effects for an NME are not desired, then a BDDCS Class 2 compond that is a substrate for brain efflux transporters would be preferable to a highly soluble BDDCS Class 1 NME. On the other hand, sponsors should recognize that if the NME was, in fact, BDDCS Class 1, then brain distribution and potential central off-target effects can not be avoided even if the compound is shown to be a substrate for brain efflux transporters.

BDDCS classification may also allow predictions regarding food effects for orally dosed drugs. The area under the curve (AUC) and bioavailability of many drugs are greatly affected by concomitant food intake and the FDA recommends that high fat meals (800–1000 cal; 50–65% from fat, 25–30% from carbohydrates, 15–20% from protein) may be used in food effect studies in humans38. Many factors are believed to contribute to these food effects, including changes in gastric emptying time, bile flow, pH of the intestine, splanchnic blood flow, and gut wall metabolism. A variety of evidence exists supporting food effects on transporters as well, as described by Custodio et al. In general high fat meals have no effect on the extent of absorption of BDDCS Class 1 drugs, increase AUC for BDDCS Class 2 drugs, decrease AUC for BDDCS Class 3 drugs, with insufficient data to show a general trend for Class 4 drugs. However, the multiplicity of factors affecting absorption in the presence of food make it more difficult to have a uniform response, and I estimate that the accuracy of the food effect predictions above is only approximately 70%.
SUMMARY
It is now been 17 years since Amidon et al. published the theoretical basis for the BCS. According to ISI this paper has now been referenced more than 1,350 times and is the most highly cited paper in the pharmaceutical sciences. The impact of this paper has been substantial leading to an FDA guidance1 and as befits the high citation rate, the paper has led to many other discoveries in the pharmaceutical sciences. Today all new drugs approved by the FDA and the EMA contain information related to the BCS classification of the molecule and its drug product. Although the purpose of BCS is to characterize drugs for which products of those drugs may be eligible for a biowaiver of in vivo bioequivalence studies, there is no inherent predictability of this characteristic, since the categorization is based on experimental results. This simple categorization of drugs into four classes has spawned many further approaches in drug product analysis. BDDCS is one of those derivative approaches. Wu and Benet recognize that the great majority of BCS Class 1 and 2 drugs were eliminated in humans predominantly by metabolic processes, while the great majority of BCS Class 3 and Class 4 drugs were predominantly eliminated unchanged either in the urine or bile. In the seven years since the BDDCS publication, it has also received a significant citation history, now close to 350. BDDCS was developed with the purpose of predicting drug disposition and drug-drug interactions, both for drugs on the market and NMEs. Very recently, Professor Amidon has characterized the 2005 BDDCS paper as advancing and conceptualizing “a new era of molecular ADMEâ€. He has very graciously written, “I believe this 2005 BDDCS paper is a seminal concept in development in the field of molecular ADME of drugs and will have a profound impact on drug discovery and development in the 21st century, and, eventually human health and quality of life.â€. I certainly hope that he is correct and in this commentary have attempted to lay out the significant role that BCS has played in drug development and the potential for the role that BDDCS may play.
Acknowledgments
I am very appreciative of the outstanding students, postdoctoral scholars and scientific collaborators who have worked with me over the years in developing the concepts of BDDCS and its role in drug development as presented here, as well as Professor Amidon and his coauthors who provided the basis for our work. Dr. Benet was supported in part in the preparation of this commentary by NIH Grant GM-061390.
Abbreviations used
| BCS | biopharmaceutics classification system |
| BDDCS | biopharmaceutics drug distribution classification system |
| EMA | European Medicines Agency |
| OATP | organic anion transporting polypeptide |
| NME | new molecular entity |
| PAMPA | parallel artificial membrane permeability assay |
| AUC | area under the curve |
 RSS Feed
RSS Feed
